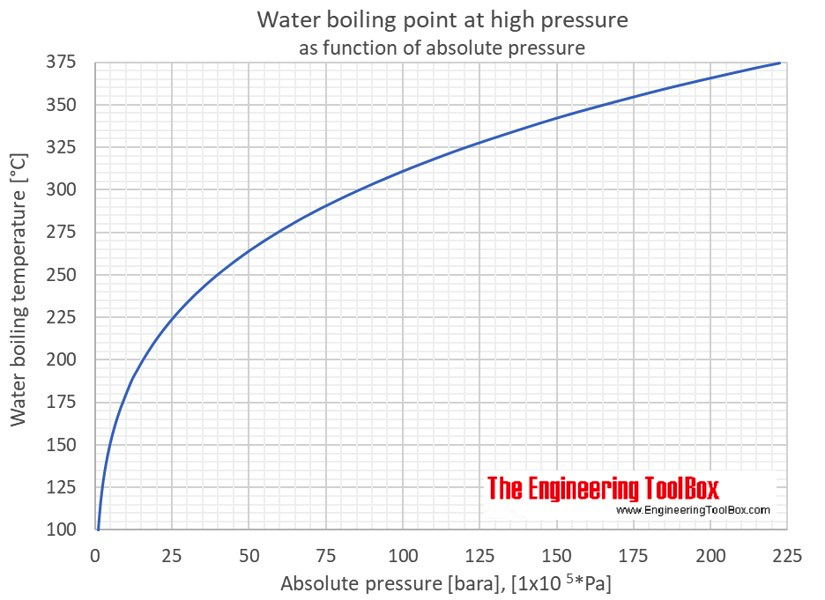
Absolute pressure = Gauge pressure + Atmospheric pressure (Atmospheric pressure at 1,01325 bar, i.e. Normal atmospheric pressure on the sea level at 0°C). Boiling point: Temperature of saturated vapour or also of ebullient water under the same pressure.
Find the mole fraction of your soIvent.The last thing we require to do before we can solve is certainly to discover the mole fraction of our solvent. Obtaining mole fractions is certainly simple: just transform your parts to moles, after that discover what percentage of the complete number of moles in the element each component occupies. In additional terms, each component's mole fraction equals(moles of component)/(total quantity of moles in the substance.)- Let's say that our formula for easy syrup utilizes1 liter (M) of water ánd 1 liter of sucrose (glucose.)In this situation, we'll need to discover the number of moIes in each. Tó do this, we'll discover the mass of each, then use the chemical's molar plenty to transform to moles.
- Mass (1 T of water): 1,000 grams (h)
- Mass (1 L of fresh sugar): Approx. 1,056.7 gary the gadget guy7
- Moles (water): 1,000 grams × 1 mol/18.015 h = 55.51 moles
- Moles (sucrose): 1,056.7 h × 1 mol/342.2965 gary the gadget guy = 3.08 moles (notice that you can discover sucrose't molar mass from its chemical formula, M12L22U11.)
- Complete moles: 55.51 + 3.08 = 58.59 moles
- Mole portion of water: 55.51/58.59 =0.947
Thevapour pressure of wateris certainly the pressure at which water vapour is in thermodynamic equilibrium with its condensed state. At higher stresses water would condense. The water vapour pressure is the partial pressure of water vapour in any gasoline combination in balance with strong or liquefied water. As for various other materials, water vapour pressure will be a function of temp and can be identified with the Clausius-Clapeyron relationship.
| Testosterone levels, °G | T, °Y | P, kPa | G, torr | P, atm |
|---|---|---|---|---|
| 0 | 32 | 0.6113 | 4.5851 | 0.0060 |
| 5 | 41 | 0.8726 | 6.5450 | 0.0086 |
| 10 | 50 | 1.2281 | 9.2115 | 0.0121 |
| 15 | 59 | 1.7056 | 12.7931 | 0.0168 |
| 20 | 68 | 2.3388 | 17.5424 | 0.0231 |
| 25 | 77 | 3.1690 | 23.7695 | 0.0313 |
| 30 | 86 | 4.2455 | 31.8439 | 0.0419 |
| 35 | 95 | 5.6267 | 42.2037 | 0.0555 |
| 40 | 104 | 7.3814 | 55.3651 | 0.0728 |
| 45 | 113 | 9.5898 | 71.9294 | 0.0946 |
| 50 | 122 | 12.3440 | 92.5876 | 0.1218 |
| 55 | 131 | 15.7520 | 118.1497 | 0.1555 |
| 60 | 140 | 19.9320 | 149.5023 | 0.1967 |
| 65 | 149 | 25.0220 | 187.6804 | 0.2469 |
| 70 | 158 | 31.1760 | 233.8392 | 0.3077 |
| 75 | 167 | 38.5630 | 289.2463 | 0.3806 |
| 80 | 176 | 47.3730 | 355.3267 | 0.4675 |
| 85 | 185 | 57.8150 | 433.6482 | 0.5706 |
| 90 | 194 | 70.1170 | 525.9208 | 0.6920 |
| 95 | 203 | 84.5290 | 634.0196 | 0.8342 |
| 100 | 212 | 101.3200 | 759.9625 | 1.0000 |
- 1Approximation formulas
Approximation formulasedit
There are many released approximations for calculating condensed vapour pressure over water and over snow. Some of these are (in approximate purchase of increasing accuracy):
- wherePwill be the vapour pressure in mmHg andTcan be the heat in kelvins.
- The Antoine formula
- where the heatTis usually in degrees Celsius (°C) and the vapour pressureGis usually in mmHg. The constants are usually provided as
| A | C | G | Testosterone levelsmin, °C | Capital tpotential, °C |
|---|---|---|---|---|
| 8.07131 | 1730.63 | 233.426 | 1 | 99 |
| 8.14019 | 1810.94 | 244.485 | 100 | 374 |
- The August-Roche-Magnus (or Magnus-Tetens or Magnus) formula, as explained in Alduchov and Eskridge (1996).2Formula 21 in2provides the coefficients used here. Discover also discussion of Clausius-Clapeyron approximations utilized in meteorology and climatology.
where heat rangeTestosterone levelscan be in °D and vapour pressureGwill be in kilopascals (kPa)
- The Tetens formula
where heatCapital tis usually in °M andGis certainly in kPa
- The Dollar formula.
whereTestosterone levelsis definitely in °Chemical andPis in kPa.
- The Goff-Gratch (1946) formula.3
Precision of various formulationsedit
Right here will be a assessment of the accuracies of these various explicit formulations, showing vividness vapour pressures for liquefied water in kPa, computed at six temperatures with their percent error from the table beliefs of Lide (2005):
| Testosterone levels(°D) | P(Lide Desk) | G(Eq 1) | G(Antoine) | P(Magnus) | P(Tetens) | G(Dollar) | P(Goff-Gratch) |
|---|---|---|---|---|---|---|---|
| 0 | 0.6113 | 0.6593 (+7.85%) | 0.6056 (-0.93%) | 0.6109 (-0.06%) | 0.6108 (-0.09%) | 0.6112 (-0.01%) | 0.6089 (-0.40%) |
| 20 | 2.3388 | 2.3755 (+1.57%) | 2.3296 (-0.39%) | 2.3334 (-0.23%) | 2.3382 (+0.05%) | 2.3383 (-0.02%) | 2.3355 (-0.14%) |
| 35 | 5.6267 | 5.5696 (-1.01%) | 5.6090 (-0.31%) | 5.6176 (-0.16%) | 5.6225 (+0.04%) | 5.6268 (+0.00%) | 5.6221 (-0.08%) |
| 50 | 12.344 | 12.065 (-2.26%) | 12.306 (-0.31%) | 12.361 (+0.13%) | 12.336 (+0.08%) | 12.349 (+0.04%) | 12.338 (-0.05%) |
| 75 | 38.563 | 37.738 (-2.14%) | 38.463 (-0.26%) | 39.000 (+1.13%) | 38.646 (+0.40%) | 38.595 (+0.08%) | 38.555 (-0.02%) |
| 100 | 101.32 | 101.31 (-0.01%) | 101.34 (+0.02%) | 104.077 (+2.72%) | 102.21 (+1.10%) | 101.31 (-0.01%) | 101.32 (0.00%) |
A more detailed debate of precision and considerations of the inaccuracy in temperatures measurements is certainly offered in Alduchov and Eskridge (1996). The analysis here displays the basic unattributed formula and the Antoine formula are fairly accurate at 100 °Chemical, but quite bad for lower temperatures above cold. Tetens can be much more precise over the range from 0 to 50 °Chemical and really aggressive at 75 °M, but Antoine's is usually excellent at 75 °C and over. The unattributed formulation must have got zero error at around 26 °M, but can be of really poor precision outside a very narrow range. Tetens' equations are usually generally much more accurate and arguably simpler for make use of at everyday temperatures (y.h., in meteorology). As anticipated, Dollar's equation forTestosterone levelsgt; 0 °D is considerably more precise than Tetens, and its brilliance increases substantially above 50 °G, though it is certainly more complex to use. The Dollar equation is definitely even excellent to the even more complicated Goff-Gratch equation over the variety required for practical meteorology.
Statistical approximationsedit
For severe calculation, Lowe (1977)4created two sets of equations for temps above and below freezing, with various amounts of precision. They are all really accurate (likened to Clausius-Clapeyron and the Goff-Gratch) but make use of nested polynomials for extremely efficient computation. However, there are more latest evaluations of probably excellent formulations, especially Wexler (1976, 1977),56documented by Flatau et al. (1992).7
Graphical pressure dependency on heatedit
Vapour pressure diagrams of water; information used from Dortmund Data Bank. Graphics shows triple point, critical stage and cooking food stage of water.
Discover alsoedit
Sourcesedit
- ^Lide, James Ur., ed. (2004).CRC Handbook of Hormone balance and Physics,(85tl ed.). CRC Press. pp. 6-8. ISBN978-0-8493-0485-9.
- ^acAlduchov, O.A.; Eskridge, R.Age. (1996). 'Improved Magnus form approximation of saturation vapor pressure'.Diary of Applied Meteorology.35(4): 601-9. Bibcode:1996JApMe.35.601A. doi:10.1175/1520-0450(1996)035lt;0601:IMFAOSgt;2.0.CO;2.
- ^Goff, L.A., and Gratch, H. 1946. Low-pressure attributes of water from −160 to 212 °F.InTransactions of the Us Culture of Heating system and Ventilating Technicians, pp 95-122, introduced at the 52nm annual conference of the Us Modern society of Heating and Ventilating Technicians, New York, 1946.
- ^Lowe, P.L. (1977). 'An approximating polynomial for the calculation of saturation vapor pressure'.Journal of Applied Meteorology.16(1): 100-4. Bibcode:1977JApMe.16.100L. doi:10.1175/1520-0450(1977)016lt;0100:AAPFTCgt;2.0.CO;2.
- ^Wexler, A new. (1976). 'Vapor pressure formulation for water in range 0 to 100°G. A revision'(PDF).J. Res. Natl. Bur. Stand up.80A(5-6): 775-785. doi:10.6028/jres.080a.071.
- ^Wexler, A new. (1977). 'Vapour pressure formula for glaciers'(PDF).L. Ers. Natl. Bur. Have.81A(1): 5-20. doi:10.6028/jres.081a.003.
- ^Flatau, G.M.; Walko, L.T.; Cotton, W.Ur. (1992). 'Polynomial fits to saturation vapour pressure'.Log of Applied Meteorology.31(12): 1507-13. Bibcode:1992JApMe.31.1507F. doi:10.1175/1520-0450(1992)031lt;1507:PFTSVPgt;2.0.CO;2.
More reading throughedit
- 'Thermophysical properties of seawater'.Matlab, EES and Excel VBA collection routines. MIT. 20 Feb 2017.
- Garnett, Pat; Anderton, Mark N; Garnett, Pamela J (1997).Chemistry Laboratory Manual For Senior Secondary School. Longman. ISBN978-0-582-86764-2.
- Murphy, M.Meters.; Koop, Capital t. (2005). 'Review of the vapour pressures of ice and supercooled water for atmospheric programs'.Quarterly Diary of the Royal Meteorological Modern society.131(608): 1539-65. Bibcode:2005QJRMS.131.1539M. doi:10.1256/qj.04.94.
- Speight, Wayne H. (2004).Lange'beds Guide of Hormone balance(16tl ed.). McGraw-Hil. ISBN978-0071432207.
Exterior linksedit
- Sixth is vömel, Holger (2016). 'Vividness vapor pressure formulations'. Boulder Company: Earth Observing Laboratory, National Middle for Atmospheric Analysis. Archived from the original on Summer 23, 2017.
- 'Vapor Pressure Calculator'. Country wide Weather Program, State Oceanic and Atmospheric Management.
Gathered from 'https://en.wikipedia.org/w/index.php?title=Vapourpressureofwateramp;oldid=895244479'
